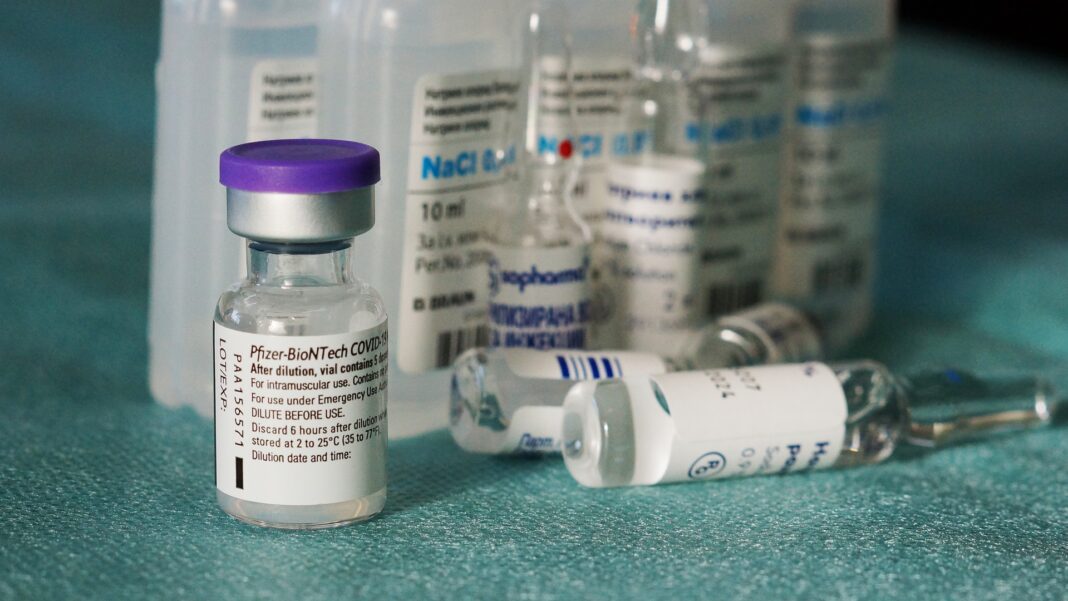
Pfizer’s mRNA-based vaccine candidate shows initial evidence of protection against Covid-19, the American pharmaceutical company announced today. Preliminary data from the phase 3 vaccine trials, which began on July 27, indicates the vaccine has a 90% efficacy rate in preventing Covid-19 in participants without prior evidence of Sars-Cov2 infection.
The trial is taking place in several countries, including the United States, Brazil, Argentina, and Germany, and has already enrolled more than 40,000 participants.
“Today is a great day for science and humanity. The first set of results from our Phase 3 COVID-19 vaccine trial provides the initial evidence of our vaccine’s ability to prevent COVID-19,”
Dr. Albert Bourla, Pfizer Chairman and CEO
Until now, there were 94 confirmed Covid-19 cases in the trial participants. The analysis is expected to continue until a total of 196 positive covid-19 cases have been confirmed. At that point, researchers also hope to answer the question of whether the mRNA-based vaccine protects against re-infection and severe forms of the disease. And, because it is only months since the first volunteers have received the vaccine, it is still impossible to say how long the protection lasts.
Pfizer and BioNtech, the German vaccine manufacturer, plan to request an Emergency Use to the Food and Drug Administration (FDA). However, such a request will occur only after the third week of November, after at least half of the patients are watched for two months for any safety issues.
Source: Press release, 09/11/2020